Electrochemotherapy (ECT) is a local cancer treatment that, in addition to its direct cytotoxic effects, also exhibits important immunomodulatory properties. By inducing immunogenic cell death, ECT can trigger an antitumor immune response, making it a promising therapeutic approach for combination with immunotherapy. Among immunotherapeutic strategies, immune checkpoint inhibitors such as antibodies targeting the PD-1 receptor play a key role in the reactivation of exhausted T lymphocytes; however, their efficacy remains limited, particularly in immunologically “cold” tumors.
In a preclinical study by Omerzel et al., we investigated the antitumor efficacy of combining bleomycin-based ECT with PD-1 blockade in four murine tumor models with distinct immune profiles: WEHI 164 fibrosarcoma, CT26 and MC38 colorectal carcinoma, and 4T1 breast carcinoma. The aim of the study was to evaluate the therapeutic effect of the combined treatment, changes in the tumor immune microenvironment, and the ability to induce long-term antitumor immune memory.
The results showed that ECT as monotherapy was most effective in immunologically “hot” tumors, WEHI and CT26, where high complete response rates were achieved. In contrast, the efficacy of ECT was limited in immunologically “cold” tumors, 4T1 and MC38. The addition of PD-1 blockade significantly enhanced the antitumor effect in the less responsive models, as reflected by a statistically significant delay in tumor growth and markedly higher complete response rates.
Immunohistochemical analysis of tumor tissues revealed increased infiltration of CD4⁺ and CD8⁺ T lymphocytes and granzyme B–positive effector cells following the combined treatment, particularly in MC38 tumors. Moreover, the formation of tertiary lymphoid structures was observed in some tumors after combination therapy, indicating localized and organized immune activation. Importantly, the combination of ECT and PD-1 inhibition also promoted the development of long-term immune memory.
Our findings confirm that ECT acts as an effective form of in situ vaccination, enabling the release of tumor antigens and the recruitment of immune cells into the tumor microenvironment, while PD-1 blockade further enhances T-cell effector function and contributes to the establishment of durable antitumor immune memory. This study highlights the importance of tailoring combination therapeutic approaches according to the immune status of the tumor and supports the further development of ECT combined with immunotherapy as a personalized strategy for cancer treatment.
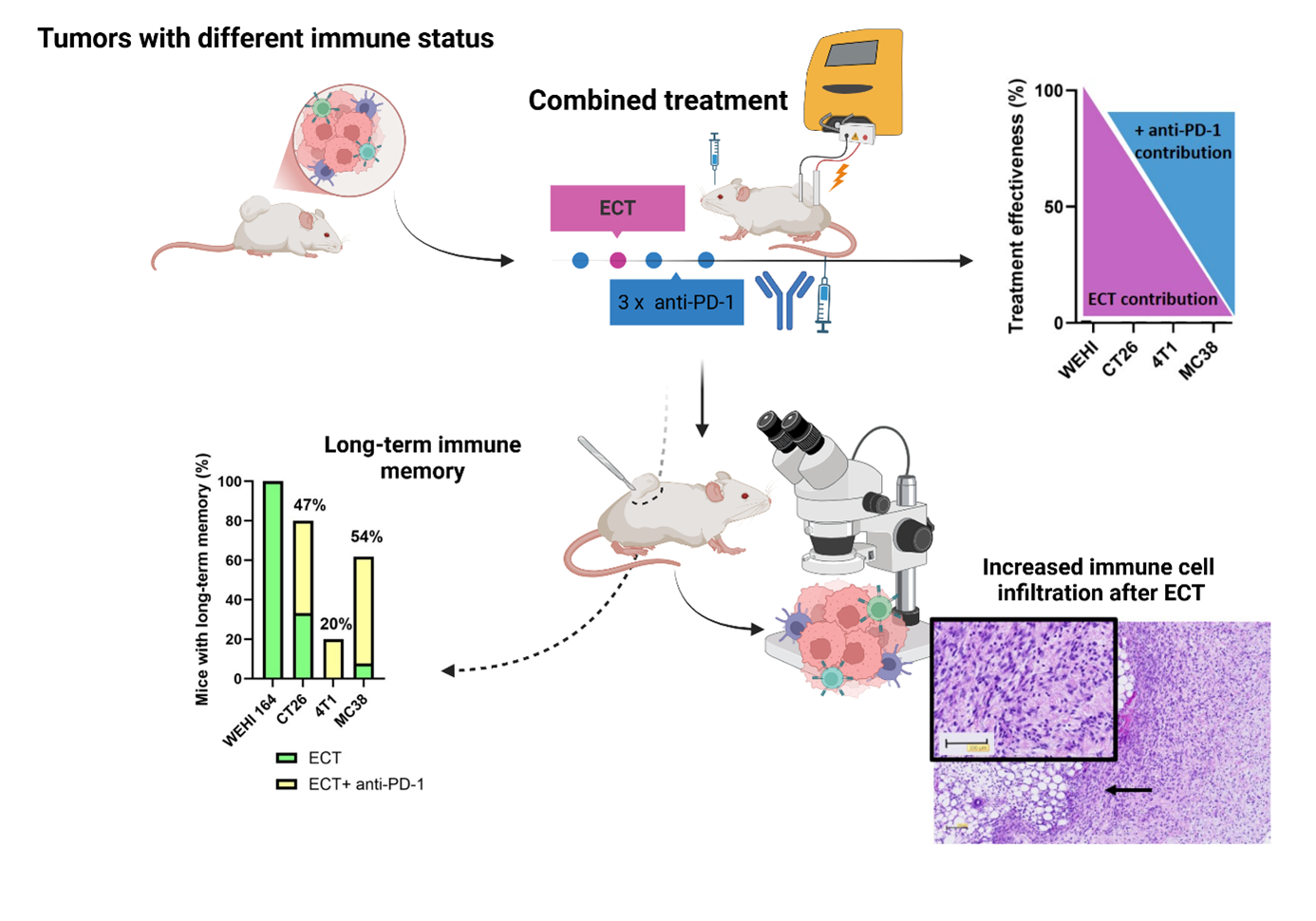
The schematic illustrates the course of the combined ECT and anti–PD-1 therapy and summarizes the key findings.
The full article is available at the following link:
