Anaphylaxis is an acute, life-threatening hypersensitivity reaction whose variability and unpredictability pose major challenges in both clinical practice and scientific research. Despite numerous studies, a comprehensive understanding of its pathogenesis remains incomplete. However, with the advancement of high-throughput technologies in the omics fields – including genomics, metagenomics, epigenomics, transcriptomics, proteomics, and metabolomics – new opportunities are emerging for an integrated, multi-layered analysis of the disease, known as the multi-omics approach.
In our recently published article, we reviewed all relevant studies from the past 25 years that have investigated individual omics layers in relation to anaphylaxis. The goal of this review was to synthesize their findings, highlight key molecular mechanisms, and identify potential biomarkers for early detection as well as for safer and more effective treatment of anaphylaxis.
The analysis of over one hundred studies revealed that numerous factors contribute to the development and progression of anaphylaxis. Genomic studies have identified gene variants associated with an increased risk of severe systemic reactions. Epigenomic research has uncovered changes in DNA methylation that may influence gene expression and modulate the intensity of immune responses. Transcriptomic analyses demonstrated a rapid activation of genes involved in innate immunity during anaphylaxis, including signaling pathways that lead to the release of inflammatory mediators, which could serve as diagnostic markers. Additionally, findings from metagenomic and metabolomic studies suggest that both the composition of the microbiome and an individual’s metabolic profile may affect susceptibility to anaphylaxis.
Collectively, these findings indicate that anaphylaxis results from a complex and interconnected interplay across all omics levels. The study highlights the need for integrated multi-omics research approaches to help identify distinct clinical subtypes (endotypes) of anaphylaxis. Such approaches are crucial for ensuring appropriate treatment and more accurate risk prediction in the future.
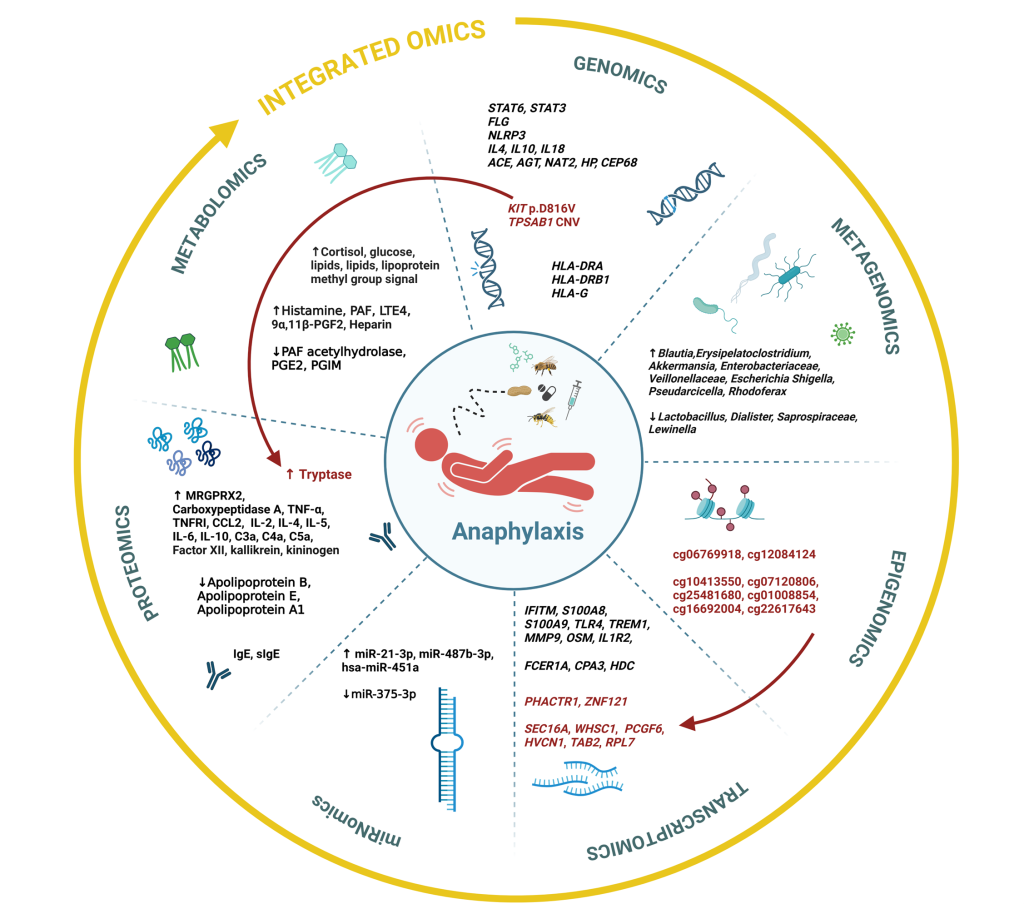
Figure: Findings across various omics levels and established associations between single-omics datasets in anaphylaxis.
URL ADDRESS:
https://link.springer.com/article/10.1007/s12016-025-09069-8
