Chemokines are small signaling proteins naturally present in the human body that regulate the migration of immune cells. Because of this function, they represent one of the promising strategies for activating immune responses within the tumor microenvironment. In particular, inflammatory chemokines have the ability to attract effector antitumor immune cells. Increased expression of inflammatory chemokines, including chemokine CCL5, could therefore enhance the infiltration of effector immune cells into tumors, which is crucial for the success of cancer immunotherapies.
In the translational study by Božič et al, we investigated a novel cancer treatment approach that combines CCL5 chemokine gene therapy with radiotherapy and evaluated the antitumor effects of the combined therapy in mouse models of colorectal cancer CT26 and breast cancer 4T1. In experiments at both the cellular and animal levels, we demonstrated that tumor cells in which CCL5 expression was increased through gene electrotransfer of plasmid DNA encoding CCL5 attracted effector immune cells. In addition, following the combined therapy, we identified changes in the expression of other inflammatory cytokines in tumors that are important for immune responses.
To assess antitumor efficacy, we tested two different doses of plasmid DNA in combination with either single-dose or fractionated radiotherapy. A statistically significant longer tumor growth delay compared with control and control plasmid DNA group was observed after the combined therapy in the breast cancer mouse model 4T1, indicating the potential of this therapeutic approach.
Graphical abstract of the article
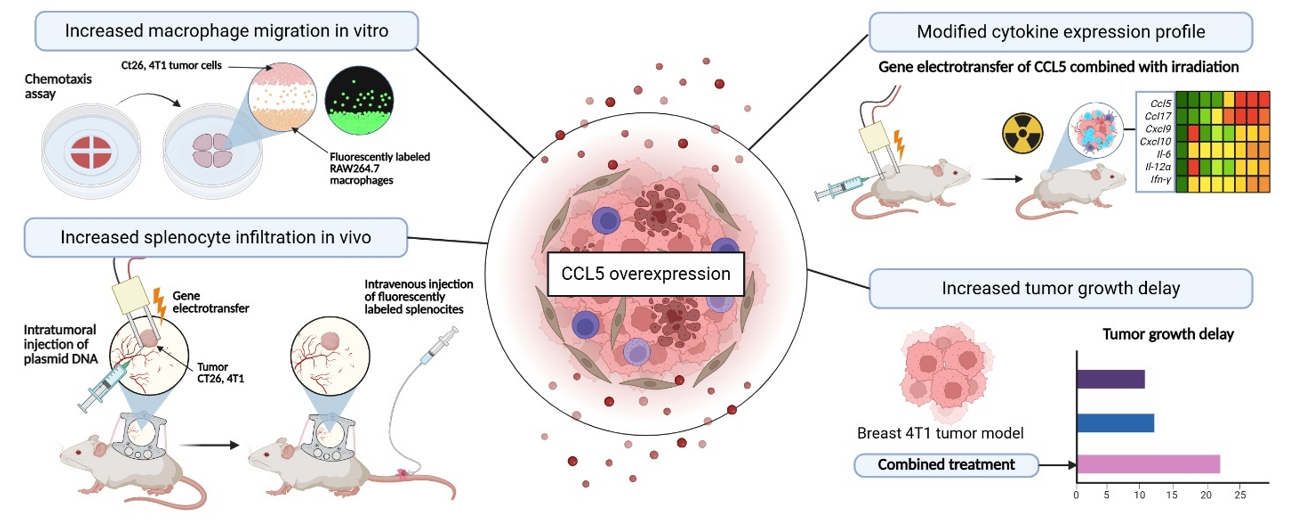
The schematic illustrates the methods used to evaluate the combined therapy of chemokine CCL5 gene electrotransfer and radiotherapy and summarizes the key findings.
The full article is available at the following link:
https://www.sciencedirect.com/science/article/pii/S0753332225010819
